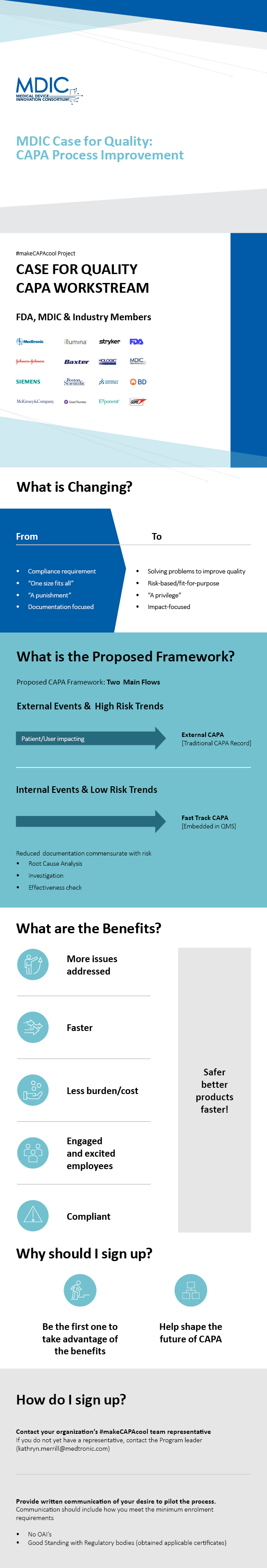
Amazon.com: Principles of corrective Action and Preventive Action :CAPA: A Handbook for Quality Professionals in Medical device and Pharmaceutical Industries eBook : MUCHEMU, DAVID N.: Kindle Store

Amazon.com: Principles of corrective Action and Preventive Action :CAPA: A Handbook for Quality Professionals in Medical device and Pharmaceutical Industries eBook : MUCHEMU, DAVID N.: Kindle Store

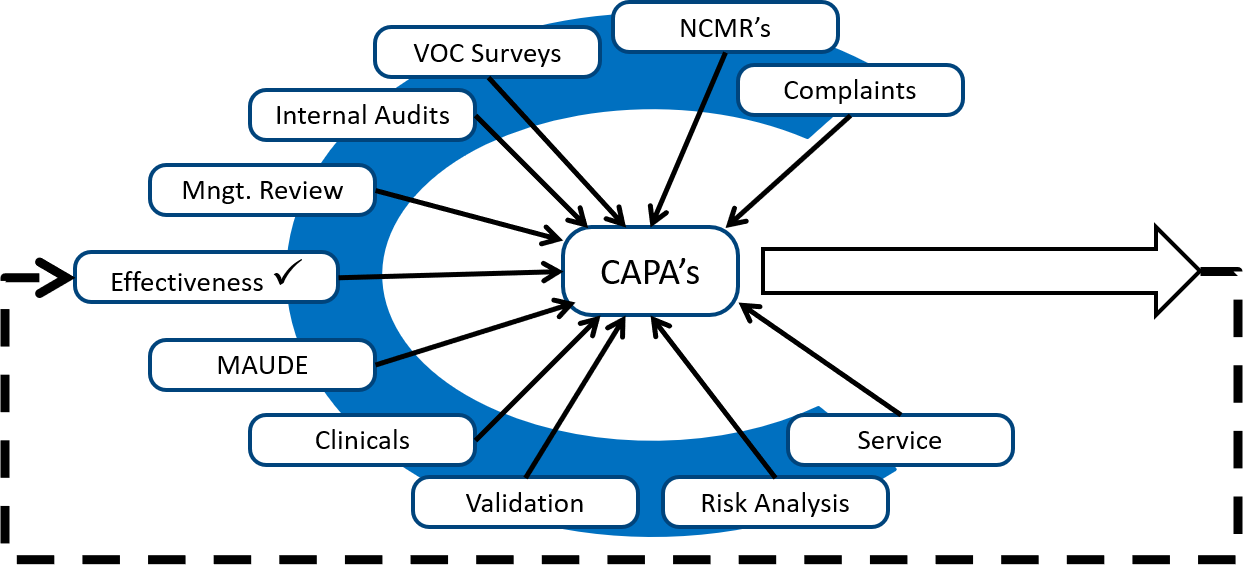
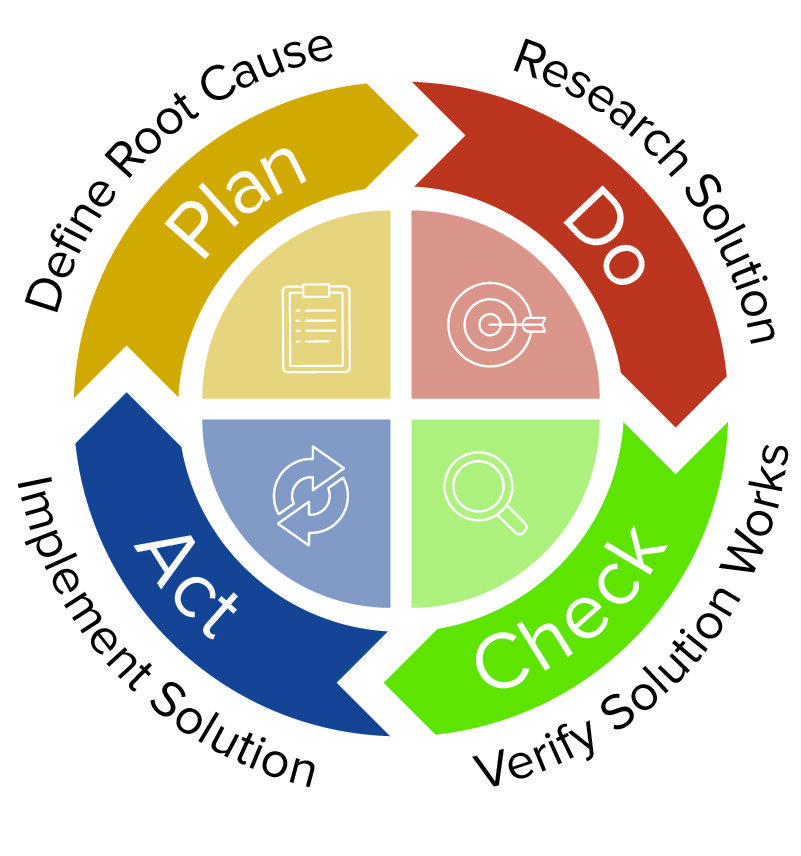
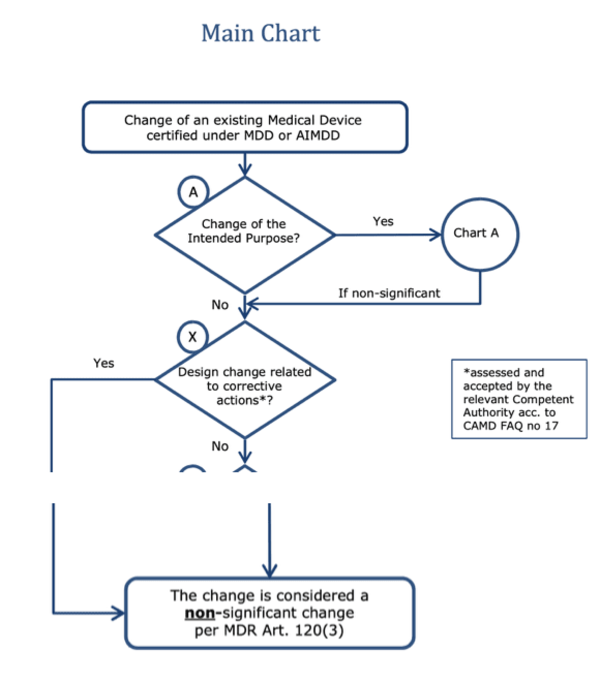
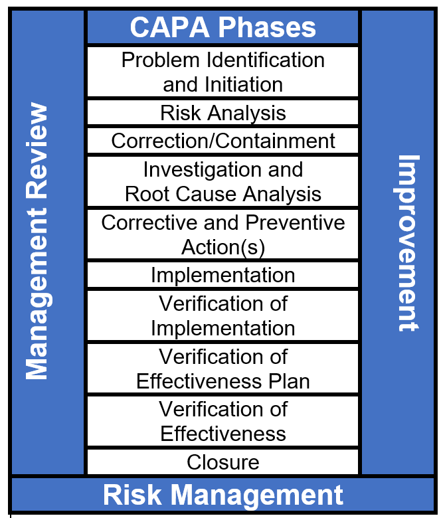
CAPA: Corrective and Preventive Actions - Introductory Principles - ALM Software Tool – Orcanos Software – ALM And Quality Management